2-Iminothiolane
Description
| 2-Iminothiolane hydrochloride (Traut's Reagent) is a useful reagent for converting primary amine functional groups into free sulfhydryl groups. Traut's reagent (2-IT) is a simple water soluble, ringed molecule that completes this primary amine (-NH2) to sulfhydryl (-SH) conversion reaction in a one-step procedure in the pH range of 7 to 10. Once modification of a protein molecule with 2-Iminothiolane is complete, free amine groups will have been irreversibly converted to a sulfhydryl group with a four atom chain and without significant disruption of the original amine groups protonation state.
After incorporation of the Traut's free sulfhydryl into a protein or otherwise, this -SH group makes for a very useful handle for further modification, such as selectively reacting this modified protein with a SMCC or BMPS containing conjugates, creating a permanent bond between two proteins. A 2-Iminothiolane modified protein or peptide could also be used to create a reversible link with an SPDP containing conjugate. Please feel free to contact our technical support staff for further questions specifically related to your application.
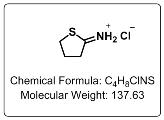
Package Sizes: 500 mg and 1 gram Product Numbers: 14104-500mg and 14104-1g Product Name: 2-Iminothiolane Alternative Names: 2-IT; Traut's Reagent; 4781-83-3; 2-Iminothiolane HCl; 2-Iminothiolane hydrochloride; Dihydro-2-(3H)-thiophenimine hydrochloride; Dihydro-2-(3H)-thiophenimine HCl; C4H8ClNS Specifications Appearance: White Solid Purity: is ≥ 95% Storage: 2 - 8 °C, protected from moisture
Properties Physical State: White Solid, Free of Foreign Matter CAS#: 4781-83-3 Chemical Formula: C4H8ClNS Molecular Weight: 137.63 Spacer length: 8.1 Å Melting Point: 197-201 °C (Lit) Tariff Code: 2934.99.9000
|