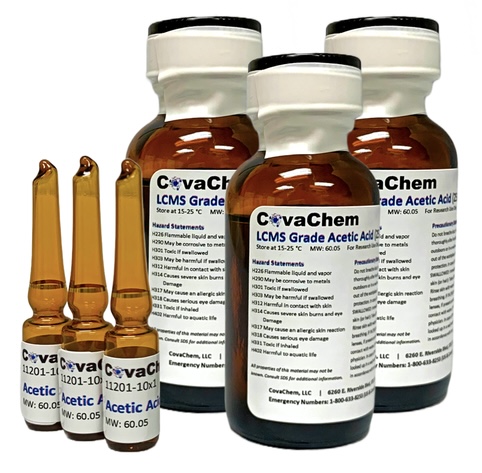
LCMS Grade Acetic Acid (UltraPure)
Description
|
LC/MS Grade Acetic Acid Product Description CovaChem's LCMS Grade Acetic Acid is ideally suited as a chromatography additive for use in both HPLC and LCMS applications. Acetic acid is typically beneficial as a separations enhancer, and has a weak ion pairing effect in the mass spectrometer, making dissociation of from the analytes relatively easy, often resulting in increased ionization efficiencies. CovaChem's LCMS Grade Acetic Acid comes in two convenient package sizes (10 x 1 mL Ampules and 25 mL bottles). The 1 mL Ampules of LCMS Grade Acetic Acid are useful for quickly preparing 1 Liter bottles of 0.1% Acetic Acid solutions in either water or other organic solvents . Product Information Package Sizes: 10 x 1 mL and 25 mL Product Numbers: 11201-10x1 or 11201-25 Product Name: LCMS Grade Acetic Acid Alternative Names: LCMS Acetic Acid; Glacial Acetic Acid; Ethanoic Acid; Methanecarboxylic acid 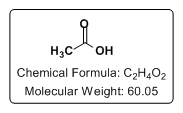
Appearance: Clear liquid Purity: is ≥ 99.8% Evaporative Residue: < 8 ppm Color Test: 10 APHA Identity: Passes Test Heavy Metals: ≤ 300 ppb Chlorides: ≤ 1.0 ppm Sulfates: ≤ 1.0 ppm Nickel: ≤ 100 ppb Cadmium: ≤ 100 ppb Iron (Fe): ≤ 200 ppb Absorbance (@350 nm): ≤ 0.02 AU Proteases: None Detected Reductant Assay: None Detected
Storage: Room Temperature Properties Physical State: Liquid CAS #: 64-19-7 Chemical Formula: C2H4O2 Molecular Weight: 60.05 amu pKa: 4.76 Boiling Point: 118-119 °C (Lit) Density: 1.049 g/mL Tariff Code: 2915.21.0000 Additional Product Information: |